 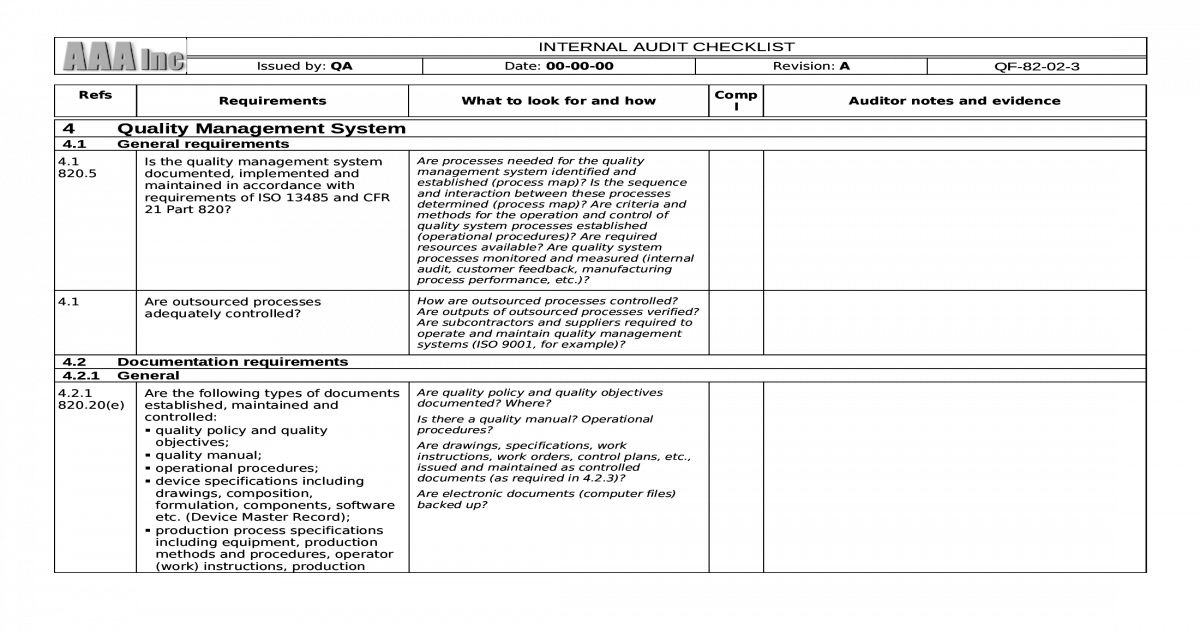
How do organization should create ISO 13485 audit checklist?Īn internal audit is there to witness the outcome of a process through a review of records or the attestation of employee actions. 
ISO 13485 audit checklists is one of the most important checking tools in a Quality Management System (QMS) for medical devices, or any management system, is the internal audit. Although it is difficult to meet the many requirements contained in the standard, an ISO 13485 audit checklist can simplify the process. Conformance with this important standard is confirmed by a complex auditing and certification process. ISO 13485:2016 is the standard for organizations that design and manufacture medical products, devices and components. All over the world, medical device manufacturers and their suppliers must meet the highest quality assurance regulations and standards such as ISO 13485. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
